Friday, July 16, 2021
Friday, February 5, 2010
Updates Iran pharma industry
Registered products: 3,700
Locally produced: 2,840
Imported : 860
Local finished product Mfg Co: 70
Local API Mfg Co: 45
Importers: 75
Pharmacy shops : 9,000
Centers for importation prior to registration:20
Locally produced: 2,840
Imported : 860
Local finished product Mfg Co: 70
Local API Mfg Co: 45
Importers: 75
Pharmacy shops : 9,000
Centers for importation prior to registration:20
Top 20 drug molecules (1st 6 month of this Iranian year)
Product Name Sales Rials 6 Month all forms all companies
INTERFERON 423,996,630,000
CEFIXIME 397,896,015,000
AMOXICILLIN 323,624,414,264
CO-AMOXICLAV 231,047,948,600
ACETAMINOPHEN 210,953,849,492
ALBUMIN 210,632,806,000
DOCETAXEL 182,924,555,600
IMIPENEM+CILASTATAIN 181,099,640,000
CLOPIDOGREL 169,915,498,015
IMMUNE GLOBULIN 160,575,717,000
ENOXAPARIN 145,486,724,600
CEFALEXIN 142,499,418,000
DEXTROSE 141,272,434,000
ATORVASTATIN 132,514,275,600
PANTOPRAZOL 130,971,241,400
SALMETEROL 127,780,028,000
SODIUM CHLORIDE 126,043,613,100
IBUPROFEN 125,621,562,500
RITUXIMAB 118,690,006,000
ERYTHROPOIETIN 110,191,720,000
INTERFERON 423,996,630,000
CEFIXIME 397,896,015,000
AMOXICILLIN 323,624,414,264
CO-AMOXICLAV 231,047,948,600
ACETAMINOPHEN 210,953,849,492
ALBUMIN 210,632,806,000
DOCETAXEL 182,924,555,600
IMIPENEM+CILASTATAIN 181,099,640,000
CLOPIDOGREL 169,915,498,015
IMMUNE GLOBULIN 160,575,717,000
ENOXAPARIN 145,486,724,600
CEFALEXIN 142,499,418,000
DEXTROSE 141,272,434,000
ATORVASTATIN 132,514,275,600
PANTOPRAZOL 130,971,241,400
SALMETEROL 127,780,028,000
SODIUM CHLORIDE 126,043,613,100
IBUPROFEN 125,621,562,500
RITUXIMAB 118,690,006,000
ERYTHROPOIETIN 110,191,720,000
Wednesday, December 10, 2008
Local Production vs Importation
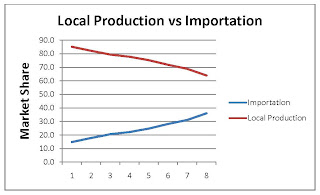
The market share of local production (value-wise) has declined from 85.2% to 63.4% over the past 8 (Iranian) years. In this period the value of importation has jumped from 14.8% to 36.6%.
It is expected that this trend - although at a much lower speed - will continue to last at least for the 5 coming years. There is also other idea that as result of global recession and low oil prices the budget for importation of pharmaceutical will be limited by Iranian government and the government will pursue the objective of fulfilling the local demand for pharmaceuticals by mainly cheap locally produced products.
Thursday, December 4, 2008
6 Months Sales
The sales of pharmaceuticals in the course of 6 first months of Iranian year (20 March 2008 to 21 September 2008) totalled to 10,799,000,000,000 Iranian Rials equal to 1,66,000,000 Mln USD (at exchange rate of 1 USD = 10,124 Iranian Rials).
The total quantity of sales of all dosage forms was 14,816,000,000 units.
The total quantity of sales of all dosage forms was 14,816,000,000 units.
Monday, December 1, 2008
Updates 1 Dec 08
Number of registered products in Iranian national formulary: 3,710
Number of registered products locally produced: 2,845
Number of registered products imported : 865
Number of local manufacturing companies: 70
Number of importing companies : 60
Number of pharmacy shops in Iran : above 9,000
Number of centers eligible for importation of Special Sales Access Programme (SSAP) of Iran MOH (prior to official registration of products in Iran: 20
Maximum qauantity of products allowed for each centre per year on SSAP basis: products meeting the demand of maximum 350 patients
Number of registered products locally produced: 2,845
Number of registered products imported : 865
Number of local manufacturing companies: 70
Number of importing companies : 60
Number of pharmacy shops in Iran : above 9,000
Number of centers eligible for importation of Special Sales Access Programme (SSAP) of Iran MOH (prior to official registration of products in Iran: 20
Maximum qauantity of products allowed for each centre per year on SSAP basis: products meeting the demand of maximum 350 patients
Sunday, August 24, 2008
Iran Pharmaceutical Industry
Iran’s Ministry of Health has a mission to provide access to sufficient quantities of safe, effective and high quality medicines that are affordable for the entire population. After the 1979 revolution, Iran adopted a full generic-based National Drug Policy, with local production of essential drugs and vaccines as one of the main goals. Nevertheless the generic system for pharmaceuticals have been reviewed since a few years ago allowing the importation and production on branded products.
The Iranian pharmaceutical market is characterized by relatively strong domestic industry, which meets the bulk of local demand in volume terms however, the country still relies on imports for raw materials and many specialized drugs. Iran MOH strongly supports and encourages local production of pharmaceuticals vs importation. Iran pharma market is a price sensitive market, sometimes a market of everything or nothing for importers which is moving slowly towards more privatization. Currently a mixture of generic products, local branded generic and international brand product are available in the market.
Currently around 60 pharmaceutical companies produce more than 95 per cent (quantitatively) of medicines on the market. Although over 85 per cent of the population use an insurance system to reimburse their drug expenses, the government still has to subsidize some of the pharmaceutical production/importation in order to increase affordability of medicines. One major problem in this sector, is also over consumption/prescription of various drugs in Iran, especially antibiotics.
Multinationals are present through imports, although improved intellectual property and regulatory conditions may attract some investment in local facilities. Currently, several successful cases of under-license manufacturing exist. Some Pharma companies are currently either producing their products under their licence or in the negotiation process with local partners for bringing their products to the market via contract manufacturing. These companies sought Iranian partners to take advantage of low production costs, a sizable market, and incentives currently offered by the Iranian government. On the other hand, although some multinationals are planning on setting up local manufacturing plants, Iran's unsettled political climate will remain a major deterrent to such ventures.
A number of multinational companies have their own branch office in Iran, and some are acting only through their agents. Nevertheless even in case of doing business via agents/distributors ,reputable international pharma companies prefer to have at least their own scientific office and execute/manage the marketing activities of their products themselves, as the agents in Iran lack professional marketing power and expertise, and there is big risk of non-compliance to marketing principles of the company if marketing activities was delegated to the agent.
The regulatory environment of the country is rather strict on the import of drugs and pharmaceuticals towards companies that intend to enter into the market especially for the first time. In the case of new unregistered companies, upon request of the importer, representatives of the MOHME will visit/inspect the manufacturing facility to evaluate its competency according to GMP. Once a company is registered it is still subject to frequent inspections making sure it can sustain its competence. On the whole pharmaceutical projects in Iran move quite slowly; although realization of each project, will guarantee good sales for a couple of years.
In the case of the import of new medicine, if the drug is already in the Iran pharmaceutical index/formulary, the import is subject to registration of brand name and the approval of the Ministry’s accredited laboratories. Otherwise the importer of that medicine should first undertake the process of registering the drug into the list. This process is considered as one of the bottle-necks of entering Iranian pharmaceutical market and is subject to meeting many requirements and can take up to one year. Preference is given for registration of products for local production rather than importation.
Currently, approximately 1,300 pharmaceutical products are registered in Iran’s National Formulary List, which roughly 950 of them are produced locally (at a value of approximately USD 1.3 bln) and the remainder is imported (at a value of approximately USD 0.7 bln).
The current growth rate of the value of market is approximately 20% (on USD basis) , and 5% quantitatively per annum (see below tables) ; this growth rate is projected to maintain in the upcoming five years.
Iran's large and rapidly growing population will be instrumental in driving the expansion of the pharmaceutical market as a whole, as will be the likely increase in drug prices, which are presently among the lowest in the region.


• Past 16 years from 154 mln Rials to 14,787 mln Rials
• Average growth rate on USD basis: 20% per year
Iran MOH could be regarded as one most bureaucratic MOH in the world, which requires constant attention to its authorities in order to run the business. Creating and keeping a sound relationship with health authorities needs special attention in Iran market. Iran MOH is mainly dominated by pharmacists, and pharmacists in Iran posses more share of voice in pharmaceutical business compared to physicians.
A good reputation of the company and its personnel is one of the success factors for the business in Iran pharma market. Fierce reaction and putting the company in black-list could not be excluded by Iran health authorities in cases of miss-conducts or non-compliance of the company to regulations, or even shortage of the product caused in the country due to delays in shipment.
Promotion of non-registered products should be strictly limited in Iran, though most of the multinational companies promote their non-registered products one way or another in order to pave the way for its registration. Promotion of registered products in the frame of regulations of Iran MOH (which is mainly in line with the worldwide accepted ethics for promotion of pharmaceuticals) is allowed
Iran has rather good distributing system for pharmaceutical products consisting of 4 main distributors and a number of small ones, which covers all the pharmacy shops and clinics throughout the country on frequent basis. Distribution of products requiring cold chain of distribution is possible.
DP, Red Crescent and IPI are among the oldest importers in Iran and during the past 5 years or so a lot of other importers have started their activities in Iran. Shafayab, Noor, and Peykdarou emerged a number of years ago as emergency centres for importation of products which are facing shortage in the country.
Iranian importers, customers and health authorities are relatively spoiled by excessive services of some international companies; on top of that one should consider the fact that Iranian customers and HAs are strikingly non-organized in their operations, and do not make accurate order forecasts. This added to many different uncertainties and obstacles (eg recent sections on credit lines on Iranian banks) in business environment of Iran and long and bureaucratic governmental procedures make the order handling of pharmaceuticals quite difficult in this country. Therefore it is not strange that country is facing shortage of different products every now and then.
Wednesday, August 20, 2008
Iran Pharmaceutical Sales
The sales of pharmaceuticals (imported + locally produced) in the course of last Iranian year (year 1386 = 20 March 2007 to 21 March 2008) totalled to 19,235,027,000,000 Iranian Rials equal to 2,019,425,000 US$ (at exchange rate of 1 US$ = 9,525 Iranian Rials.
The share of imported products in this year was 36% of the total sales, and 64% was accounted to the local production.
The share of imported products in this year was 36% of the total sales, and 64% was accounted to the local production.
Tuesday, August 19, 2008
Top selling products - value
The 10 to selling products (value wise) during the last Iranian year were as follows:
1-TIENAM® 500MG VIAL
2-TAXOTERE® 80MG 2ML VIAL
3-BETAFERON® 8MIU VIAL
4-REBIF® 44MCG/0.5ML SYRINGE
5-CEFTRAX® 1G VIAL
6-NORDITROPIN® 4U VIAL
7-CELLCEPT® 500MG TAB
8-GEMZAR® 1G VIAL
9-DIPHERELINE® 3.75MG VIAL
10-COSAR-AMOXICILLIN® 500MG CAP
1-TIENAM® 500MG VIAL
2-TAXOTERE® 80MG 2ML VIAL
3-BETAFERON® 8MIU VIAL
4-REBIF® 44MCG/0.5ML SYRINGE
5-CEFTRAX® 1G VIAL
6-NORDITROPIN® 4U VIAL
7-CELLCEPT® 500MG TAB
8-GEMZAR® 1G VIAL
9-DIPHERELINE® 3.75MG VIAL
10-COSAR-AMOXICILLIN® 500MG CAP
Sunday, August 17, 2008
Top selling products - quanity
10 top selling product (quantity wise) during the last Iranian year were as follows:
1-DIPHNOKIM® 2.5MG TAB
2-RANITIDINE 150MG TAB
3-ADULT COLD TAB
4-FERIRON® TAB
5-ADULT COLD TAB
6-ASA 80MG EC TAB
7-COSAR-AMOXICILLIN® 500MG CAP
8-ATENOLOL 50MG TAB
9-DIPHENOXYLATE 2.5MG TAB
10-ANTIHISTAMINE DECONGESTANT TAB
1-DIPHNOKIM® 2.5MG TAB
2-RANITIDINE 150MG TAB
3-ADULT COLD TAB
4-FERIRON® TAB
5-ADULT COLD TAB
6-ASA 80MG EC TAB
7-COSAR-AMOXICILLIN® 500MG CAP
8-ATENOLOL 50MG TAB
9-DIPHENOXYLATE 2.5MG TAB
10-ANTIHISTAMINE DECONGESTANT TAB
Saturday, August 16, 2008
Registration of Pharmaceutical Products in Iran
Documents required for registration of a pharmaceutical product in Iran after its addition to Iranian list of formulary
I- Certificate of Pharmaceutical Product according the specification of WHO
I I-A certificate/letter containing list of the countries where product (in any dosage form) is registered and their registration number
I II-A certificate/letter containing list and of the countries where product (any dosage form) is sold/used, and information regarding date of introduction in these markets.
IV-A certificate/letter has a factory/manufacturing unit in them and the address of factory containing list of the countries in which the company
V-Letter of agency for the product(s) to an Iranian company
VI-Complete form for price of product
VII-Complete Drug Importing Application
VIII-Samples of product including CoA and all analytical records
plus a comprehensive file (DMF = Drug Master File) containing following documents:
1-Company Profile
Including also (not limited to ) plant master file including also the machinery, and equipments used for production and analysis of the product.
2- Manufacturer
-Manufacturing authorization holder (name, address, country)
-Number and date of the first marketing authorization/renewal
-Manufacturer of finished product (name, address, country)
-Flow-chart indication the different sites involved in the manufacturing process, packaging and release of the medicinal product.
-Manufacturer of the active substance(s) (Name, address and country)
3-Formula/composition of the product
-Quantitative and qualitative composition in terms of the active substance(s)
-List of active substances separately form the excipient(s)
(Name of active substance (INN), name of excipients, function, quantity, unit, reference/pharmacopoeia)
-Incompatibilities of the excipients
-List of materials of animal and/or human origin contained or used in the manufacturing process of the medicinal product
-List of constitutes from other origins
-Coloring, flavoring and perfume compounds
4-Compostion of batch
-Complete composition of a trial batch
-Complete composition of batch for production at industrial level
S-Packaging material of the product
1-specification
2-method of analysis
3-certificate of analysis
* 4-Samples of packaging material
6-Method of Preparation
a-manufacturing formula
b-manufacturing process*
c-manufacturing equipments
7-Control of starting materials
a-active ingredients
1-specifications
2-method of analysis
3-potential impurities and determination
4-certificate of analysis*
b-other ingredients
1-specification
2-method of analysis
4-certificate of analysis*
5-the non-active substances not-remaining in the finished product
8-Control tests on finished products
a-specification
b-batch analysis
c-certificate of analysis*
d-the non-active substances remaining in the finished product
e-the limitation for the potentially toxic substance(s) which may remain in the finished product and the method for their determination.
9-Sampling
-Method of sampling
-Details of sampling plan
-Method of selection of samples for analysis
-Method of increasing or decreasing the number of samples
-Deviations and necessary actions needed to be taken based on statistical methods.
10-ln-process controls
-Details of physical, chemical, microbiological and other tests during the process of production
-Deviations in in-process tests and actions needed to be taken
-Statistical methods for combination of the result of tests on consecutive batches
-Method of analysis of in-process control as a feed-back for improving/adopting the production process
11-Pharmacopial specification of the product
-Acceptable range of physical, chemical, microbiological and other tests based on the relevant pharmacopeias
-Conformity of the product with specification of the pharmacopeias
-Pharmacopeias tests for guaranteeing the clinical, physical, chemical, microbiological characteristics of the product in the course of its shelf-life
-Comparisons/conformity of the release characteristics of the product
-Additional test compared to pharmacopeias specifications.
-Alternative tests to pharmacopeias tests
12-Stability
a-controlled batches
b-storage conditions
c-description of containers
d-analytical methods
e-stability schedule
f-results*
g-interpretation of results h-shelf life*
i-validation of stability tests
13-Pharmacology and Toxicology report
14-Clinical trials
15-Bioequivalency, and pharmacodynamics
16-Patient information leaflet
17 -Labels and outer carton text
1S-Samples of the product with original certificate of analysis plus the required reference standard for testing the samples
N.B:
-Documents "I" to "V" in above need to be original copy and need to be legalized by Iranian Embassy.
-Documents marked as "*" within the above list of "1" to "18" need to be an original copy and signed and stamped by the responsible person in the factory/company.
Number of samples required. Samples: -Large volume parenterals
-Small volume parenterals 1 and 2 cc -Small volume parenterals 5 to 10 cc -Drop, sterile ointment
-Oral solutions, syrups, suspensions -Topical ointment, cream, spray -Shampoo
-Tablets and capsules
-Soaps
-Suppositories
-Injectable vials
20 200 100
60 20 20 15
200
10 50 50
I- Certificate of Pharmaceutical Product according the specification of WHO
I I-A certificate/letter containing list of the countries where product (in any dosage form) is registered and their registration number
I II-A certificate/letter containing list and of the countries where product (any dosage form) is sold/used, and information regarding date of introduction in these markets.
IV-A certificate/letter has a factory/manufacturing unit in them and the address of factory containing list of the countries in which the company
V-Letter of agency for the product(s) to an Iranian company
VI-Complete form for price of product
VII-Complete Drug Importing Application
VIII-Samples of product including CoA and all analytical records
plus a comprehensive file (DMF = Drug Master File) containing following documents:
1-Company Profile
Including also (not limited to ) plant master file including also the machinery, and equipments used for production and analysis of the product.
2- Manufacturer
-Manufacturing authorization holder (name, address, country)
-Number and date of the first marketing authorization/renewal
-Manufacturer of finished product (name, address, country)
-Flow-chart indication the different sites involved in the manufacturing process, packaging and release of the medicinal product.
-Manufacturer of the active substance(s) (Name, address and country)
3-Formula/composition of the product
-Quantitative and qualitative composition in terms of the active substance(s)
-List of active substances separately form the excipient(s)
(Name of active substance (INN), name of excipients, function, quantity, unit, reference/pharmacopoeia)
-Incompatibilities of the excipients
-List of materials of animal and/or human origin contained or used in the manufacturing process of the medicinal product
-List of constitutes from other origins
-Coloring, flavoring and perfume compounds
4-Compostion of batch
-Complete composition of a trial batch
-Complete composition of batch for production at industrial level
S-Packaging material of the product
1-specification
2-method of analysis
3-certificate of analysis
* 4-Samples of packaging material
6-Method of Preparation
a-manufacturing formula
b-manufacturing process*
c-manufacturing equipments
7-Control of starting materials
a-active ingredients
1-specifications
2-method of analysis
3-potential impurities and determination
4-certificate of analysis*
b-other ingredients
1-specification
2-method of analysis
4-certificate of analysis*
5-the non-active substances not-remaining in the finished product
8-Control tests on finished products
a-specification
b-batch analysis
c-certificate of analysis*
d-the non-active substances remaining in the finished product
e-the limitation for the potentially toxic substance(s) which may remain in the finished product and the method for their determination.
9-Sampling
-Method of sampling
-Details of sampling plan
-Method of selection of samples for analysis
-Method of increasing or decreasing the number of samples
-Deviations and necessary actions needed to be taken based on statistical methods.
10-ln-process controls
-Details of physical, chemical, microbiological and other tests during the process of production
-Deviations in in-process tests and actions needed to be taken
-Statistical methods for combination of the result of tests on consecutive batches
-Method of analysis of in-process control as a feed-back for improving/adopting the production process
11-Pharmacopial specification of the product
-Acceptable range of physical, chemical, microbiological and other tests based on the relevant pharmacopeias
-Conformity of the product with specification of the pharmacopeias
-Pharmacopeias tests for guaranteeing the clinical, physical, chemical, microbiological characteristics of the product in the course of its shelf-life
-Comparisons/conformity of the release characteristics of the product
-Additional test compared to pharmacopeias specifications.
-Alternative tests to pharmacopeias tests
12-Stability
a-controlled batches
b-storage conditions
c-description of containers
d-analytical methods
e-stability schedule
f-results*
g-interpretation of results h-shelf life*
i-validation of stability tests
13-Pharmacology and Toxicology report
14-Clinical trials
15-Bioequivalency, and pharmacodynamics
16-Patient information leaflet
17 -Labels and outer carton text
1S-Samples of the product with original certificate of analysis plus the required reference standard for testing the samples
N.B:
-Documents "I" to "V" in above need to be original copy and need to be legalized by Iranian Embassy.
-Documents marked as "*" within the above list of "1" to "18" need to be an original copy and signed and stamped by the responsible person in the factory/company.
Number of samples required. Samples: -Large volume parenterals
-Small volume parenterals 1 and 2 cc -Small volume parenterals 5 to 10 cc -Drop, sterile ointment
-Oral solutions, syrups, suspensions -Topical ointment, cream, spray -Shampoo
-Tablets and capsules
-Soaps
-Suppositories
-Injectable vials
20 200 100
60 20 20 15
200
10 50 50
Tuesday, August 12, 2008
Salary Range of Multinational Pharmaceutical Companies in Iran
The below are the monthly salary range for employees with Pharm.D. or M.D. degree:
- Medical Representative: 1,000 to 1,700 US$
- Supervisor and Product Manager: 1,800 to 3,000 US$
- Sales Manager and Marketing Manager: 3,000 to 5,000 US$
- Country Manager: 5,000 to 10,000 US$
Monday, August 11, 2008
Pricing , Mark ups
The average mark-up for pharmaceuticals in Iran are as follows:
Custom Tax: 4% (for products not locally produced) and 65% ( for products which are locally produced)
Clearance/etc. expenses:4%
Importers profit: 13%
Distributors profit: 10%
Pharmacy shop profit: 15%
Custom Tax: 4% (for products not locally produced) and 65% ( for products which are locally produced)
Clearance/etc. expenses:4%
Importers profit: 13%
Distributors profit: 10%
Pharmacy shop profit: 15%
Sunday, August 10, 2008
Leading Importers
10 leading importers in last Iranian year were as follows:
1-Cobel Darou
2-Shafayab Goster
3-Jahan Behbood
4-Akbarieh
5-Behestan Darou
6-Tejarat and Sanat Group
7-KBC
8-Poura-Teb
9-Kamal Darou
10-Nedayeh Mahya
1-Cobel Darou
2-Shafayab Goster
3-Jahan Behbood
4-Akbarieh
5-Behestan Darou
6-Tejarat and Sanat Group
7-KBC
8-Poura-Teb
9-Kamal Darou
10-Nedayeh Mahya
Saturday, August 9, 2008
Leading Distributors
10 leading distributors in last Iranian year:
1-Darou Pakhsh
2-Pakhsh Razi
3-Pakhsh Hedjrat
4-Pakhsh Ferdous
5-Pakhsh Alborz
6-Pakhsh Ghasem
7-Darou Gostar Razi
8-Darou Behdasht Shafa Arad
9-Armaghan Darou
10-Mahya Darou
1-Darou Pakhsh
2-Pakhsh Razi
3-Pakhsh Hedjrat
4-Pakhsh Ferdous
5-Pakhsh Alborz
6-Pakhsh Ghasem
7-Darou Gostar Razi
8-Darou Behdasht Shafa Arad
9-Armaghan Darou
10-Mahya Darou
Wednesday, August 6, 2008
Leading Manufacturers
Iranian leading manufacturers are as follows:
1-Darou Pakhsh
2-Farabi
3-Exir
4-Jaber Ebn Hayan
5-Alborz Darou
6-Kosar
7-Tehran Shimi
8-Zahravi
9-Sina Darou
10-Sobhan Darou
1-Darou Pakhsh
2-Farabi
3-Exir
4-Jaber Ebn Hayan
5-Alborz Darou
6-Kosar
7-Tehran Shimi
8-Zahravi
9-Sina Darou
10-Sobhan Darou
Friday, August 1, 2008
Pharmacy Schools in Iran
Tehran Univeristy of Medical Sciences and Health Services
School of Pharmacy
Enghelab Square, 16 Azar Street, Tehran University, Faculty of Pharmacy
P.O.Box: 14155 - 6451, Tehran, Iran
Tel:+98 21 66959090-8
E.Mail: pharmacy@sina.tums.ac.ir
URL: http://pharmacy.tums.ac.ir/fa/home/Default.aspx
--------------------------------------------------------------------------------
Shahid Beheshti University of Medical Sciences and Health Services
School of Pharmacy
No 105, Shams Alley, Across from Tavaneer Ave., Vali-E-Asr Ave
P.O. Box: 14155 - 6153, Tehran
Tel: +98 21 8773523
Fax: +98 21 8795008
Email: pharmacy@morva.net
URL: http://pharmacy.morva.net/
--------------------------------------------------------------------------------
Shiraz University of Medical Sciences - Pharmacy School
P.O. Box: 71345-1849
Shiraz
Tel: +98
Fax: +98
Email: meddean@sums.ac.ir
URL: www.sums.ac.ir/
--------------------------------------------------------------------------------
Tabriz University of Medical Sciences - School of Pharmacy
Tabriz 21664
Tel: +98 411 334 1315
Fax: +98 411 334 4798
Email: GarjaniA@tbzmed.ac.ir
URL: www.tbzmed.ac.ir
School of Pharmacy
Enghelab Square, 16 Azar Street, Tehran University, Faculty of Pharmacy
P.O.Box: 14155 - 6451, Tehran, Iran
Tel:+98 21 66959090-8
E.Mail: pharmacy@sina.tums.ac.ir
URL: http://pharmacy.tums.ac.ir/fa/home/Default.aspx
--------------------------------------------------------------------------------
Shahid Beheshti University of Medical Sciences and Health Services
School of Pharmacy
No 105, Shams Alley, Across from Tavaneer Ave., Vali-E-Asr Ave
P.O. Box: 14155 - 6153, Tehran
Tel: +98 21 8773523
Fax: +98 21 8795008
Email: pharmacy@morva.net
URL: http://pharmacy.morva.net/
--------------------------------------------------------------------------------
Shiraz University of Medical Sciences - Pharmacy School
P.O. Box: 71345-1849
Shiraz
Tel: +98
Fax: +98
Email: meddean@sums.ac.ir
URL: www.sums.ac.ir/
--------------------------------------------------------------------------------
Tabriz University of Medical Sciences - School of Pharmacy
Tabriz 21664
Tel: +98 411 334 1315
Fax: +98 411 334 4798
Email: GarjaniA@tbzmed.ac.ir
URL: www.tbzmed.ac.ir
Monday, July 21, 2008
Razi Father of Pharmcay in Iran

Abū Bakr Muhammad ibn Zakariyā Rāzī (Arabic: أبو بكر محمد بن زكريا الرازي, Persian: زكريای رازی Zakariā-ye Rāzi; Latin: Rhazes or Rasis) was a Persian, chemist, physician, philosopher and scholar. According to Biruni, Razi was born in Rayy, Iran in the year 865 (251 AH) and died there in 925 (313 AH).
Razi made fundamental and enduring contributions to the fields of medicine, alchemy, and philosophy, recorded in over 184 books and articles in various fields of science. He was well-versed in Persian, Greek and Indian medical knowledge and made numerous advances in medicine through own observations and discoveries. He was an early proponent of experimental medicine and is considered the father of pediatrics. He was also a pioneer of neurosurgery and ophthalmology.
As an alchemist, Razi is known for his study of sulfuric acid. He also discovered ethanol and refined its use in medicine.
Razi was a rationalist, and was very confident in the power of reason; he was widely regarded by his contemporaries and biographers as liberal and free from any kind of prejudice, very bold and daring in expressing his ideas without a qualm.
He traveled extensively and rendered service to several princes and rulers, especially in Baghdad, where his laboratory was located. As a teacher in medicine, he attracted students of all disciplines and was said to be compassionate and devoted to the service of his patients, whether rich or poor.
The modern-day Razi Institute in Tehran, and Razi University in Kermanshah were named after him, and 'Razi Day' ('Pharmacy Day') is commemorated in Iran every August 27.
Subscribe to:
Posts (Atom)